
Plant-derived lipid nanoparticles: a new potential therapeutic approach for traditional Chinese medicine’s fresh herbs
Fresh herbs are unique forms of traditional Chinese medicine (TCM)
The application of fresh herbs runs through the origin and development of TCM. Ancient practitioners of TCM have a good understanding of the fresh herbs’ unique characteristics (distinct from dried herbs), which can be evidenced in many ancient medical books. More than two thousand years ago, the “Shennong Ben Cao Jing” highly praised that fresh ginger [Zingiber officinale Rosc.]’s efficacy is exceptionally better than dried ginger (1). In Eastern Han Dynasty, Zhongjing Zhang’s “Shanghan Lun” and “Jingui Yaolüe” documented fresh herbs’ applications to treat a variety of conditions. For example, the Essential Prescriptions from the Golden Cabinet, another name of the Jingui Yaolüe, recorded the use of Lily [Lilium Lanci folium Thunb; Lilium brownii F. E. Brown var. viridulum Baker; Lilium pumilum DC.] Rehmannia [Rehmannia glutinosa Libosch.] Soup, which employs lily roots and raw Rehmannia juices to nourish the atrium (heart chamber) and diminishes blood fever (2). It is also worth noting that Hong Ge’s book “A Handbook of Formulas for Emergencies” (written in the Eastern Jin Dynasty) introduced a fresh herb-based anti-malaria parasite approach: “mixing one grip of freshly collected Artemisia [Artemisia annua L.] with two liters of water and mashing to get the juice.”(3) This precious record became the source of inspiration for Professor Tu, Youyou, whose research brought the anti-malaria drug artemisinin that saved millions of lives and won her the Nobel Prize in Physiology or Medicine in 2015 (4,5).
With the prevalence of the Febrile disease theory, fresh medicines are also increasingly used in TCM prescriptions (6,7). For instance, Ziming Chen’s famous prescription “bolus of four fresh drugs” comprises fresh meshed lotus [Nelumbo nucifera Gaertn.] leaves, fresh Argy wormwood [Artemisia argyi Levl. Et Vant.] leaves, fresh cypress [Platycladus orientalis (L.) Franco] leaves, and raw Rehmannia (8). Kentang Wang’s “four-juice drink” is made from fresh grape [Vitis vinifera L.] juice, fresh lotus root juice, raw Rehmannia juice, and honey (9). In the Ming and Qing dynasties, TCM practitioners accumulated rich experience in the clinical application of fresh herbs, and the understanding of the extraordinary efficacy of fresh herbs is more in-depth. Since then, fresh herbs-based prescriptions have formed powerful TCM tools in treating diseases in clinical use (1,10).
Fresh herb-derived lipid nanoparticles (FHDLNs) are becoming new therapeutic approaches in preclinical studies
FHDLNs are exosome-like membrane vesicles released by medicinal plants, such as ginger, garlic [Allium sativum L.], and grape. FHDLNs exhibit spherical structures containing miRNAs, proteins, and secondary metabolites that were encapsulated in lipid bilayers. They are biocompatible, have therapeutic effects, and present tissue-specific targeting properties. Thus, they have great potential in clinic application as a new type of naturally occurring nanopharmaceuticals or nanoplatform for drug delivery (11).
Compared to available synthetic drug delivery systems, FHDLNs offer multiple advantages, such as higher internalization rate, lower immunogenicity, and more economical mass production. Reassembled FHDLNs (Figure 1) also have a strong capacity of carrying various therapeutic agents, including mRNAs, proteins, and small molecule drugs, mimicking their originated state that serves as extracellular exosomes to mediate cell-cell communication (12,13).
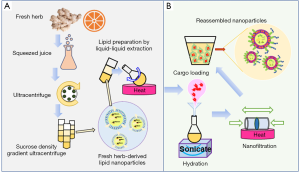
Fresh ginger-derived nanoparticles (GDNPs) target the colon and reduce colitis in animal models
GDNPs were recently isolated from freshly squeezed ginger juice (12,14). Characterization of GDNPs shows that they are spherical lipid nanoparticles with a size of ~220 nm and a surface zeta potential value of ~−12 mV. Their lipid composition analysis revealed that GDNP lipid biolayer mainly comprised phosphatidic acid (PA; ~25% to 40%), digalactosyl diacylglycerol (DGDG; ~25% to 40%), and mono-digalactosyl diacylglycerol (MGDG; ~20% to 30%).
Oral administration of a specific population of GDNPs targets the colon, which is crucial in treating colonic diseases. Researchers have further proved that orally delivered GDNPs could reduce ulcerative colitis and colitis-associated cancer in experimental mouse models. Such efficacy might attribute to GDNPs’ carrying contents, including 6-gingerol, 6-shogaol, and ginger miRNAs. RNA sequencing revealed that GDNPs contained more than 100 types of miRNAs, which may target various human genes on 3’ untranslated regions (3’UTR). GDNPs did not show noticeable in vivo toxicity (14,15). All these findings demonstrate that GDNP is an excellent fresh herb-derived nanopharmaceuticals.
Using the lipids isolated from GDNPs, scientists re-engineered ginger nanoparticle-derived lipid nanovectors (GNDLNs), which share identical nanostructures as GDNPs but only contain clearly defined lipids. GNDLNs showed negligible toxic effects at concentrations up to 100 mM, more biocompatible than commercial liposomes. These self-assembled GNDLNs can encapsulate 6-shogaol or doxorubicin to deliver them to the colon to treat ulcerative colitis or colon cancer (16-18).
Garlic-derived nanovesicles (GDNVs) target liver cells via CD98 mediated pathway
Recently, researchers isolated GDNVs from the fresh garlic and showed that the GDNVs’ surface proteins played an essential role in the particles’ endocytosis by liver cells (HepG2). They discovered that blocking GDNV II lectin (mannose-specific binding protein) or liver cells’ CD98 receptors could significantly reduce the cell uptake of GDNVs by HepG2. GDNVs can significantly downregulate pro-inflammatory factors in the HepG2 cells; they exhibited an excellent in vitro anti-inflammatory effect (19). The CD98 mediated GDNVs internalization might be a process associated with the cellular anti-inflammatory effects of garlic.
Grape exosome-like nanoparticles (GELNs) promote the growth of the intestinal stem cells
GELNs were found to share specific proteins with the mammalian-derived exosomes, including HSP70 and aquaporin proteins (13). The study showed that GELNs have unique transport properties and biological functions. Orally administrated GELNs can penetrate the intestinal mucus barrier and be taken up by mouse intestinal stem cells. They induce the replication of Lgr5+ intestinal stem cells through the Wnt/β-catenin pathway and can protect mice from dextran sulfate sodium (DSS)-induced colitis. Researchers further demonstrated that lipids isolated from GELN played a vital role in the inducing of Lgr5+ stem cells, and the reassembled lipid nanoparticles (with lipids isolated from GELNs) were necessary for in vivo targeting the intestinal stem cells. GELNs and reassembled lipid nanoparticles can modulate the stem cell-based tissue renewal process in response to pathological simulates. This finding paved the road for developing novel and safe, fresh herb-based strategies for colonic tissue regeneration.
Future of fresh herb-derived nanotherapeutics: can we put new wine in old bottles?
Produced by various fresh herbs, FHDLNs contribute to transfer molecules that can render new therapeutic properties or reprogram the recipient cells (12,15,20). Most FHDLNs were found to be able to travel along the gut, migrate through the intestinal mucus, and be taken up by the intestine (12). These FHDLNs can protect the encapsulated molecules from degradation in the gastrointestinal tract’s harsh environment, which expanded our knowledge of why and how the fresh herb-based treatment could be superior to some traditional prescriptions that only use dried or/and processed herbs. Maintaining the freshness is crucial to produce the FHDLNs as the dehydration of fresh plant increases the intracellular partition of amphiphilic compounds into the lipid phase (21), which changes the membrane lipid composition and may disable the self-assembly capability of membrane lipids into nanoparticles.
It is also proven feasible to produce and store large quantities of nanoparticles from fresh herbs, potentiating this technology into clinical settings to treat diseases such as inflammatory bowel disease, colorectal cancer, and others. However, the current technical limitation of FHDLNs is the lack of a sterile technique to ensure the stability of the lipid nanostructure and the loaded macromolecule cargos (such as siRNA or mRNA).
It is also necessary to establish a quality control method of the fresh herbal material and simplify the preparation process that can decrease the batch-to-batch variation of produced FHDLNs. As nanotechnology’s broad application only took place for less than 25 years, the accelerating research and development in FHDLNs-based disease treatment certainly require to be supported by theories from either modern pharmacology or TCM. However, the TCM’s theories, such as Yin and Yang and Five elements, can hardly reflect the latest nano pharmaceutics’ discoveries or elucidate the underlying mechanisms of the FHDLNs’ efficacy. There is an urgent need for an upgrade or refresh in the TCM theories so that TCM could embrace and keep abreast with the fast pace of the development of modern nanopharmaceuticals.
Acknowledgments
Funding: This work was supported by the Institute for Biomedical Sciences (IBMS) and the Digestive Diseases Research Group (DDRG) of Georgia State University.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Longhua Chinese Medicine. The article has undergone external peer review.
Peer Review File: Available at http://dx.doi.org/10.21037/lcm-21-5
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/lcm-21-5). CY serves as an unpaid editorial board member of Longhua Chinese Medicine from Oct 2020 to Sep 2022. The author has no conflicts of interest to declare.
Ethical Statement: The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Fang WT, Zhan ZL, Peng HS, et al. Historical evolution and change of differentiation on dried ginger, fresh ginger and baked ginger. Zhongguo Zhong Yao Za Zhi 2017;42:1641-5. [PubMed]
- Zhang Z. Essentials from the Golden Cabinet. China: Han.
- Li M, Liang Y. Ge Hong and Zhou Hou Jiu Zu Fang (A Handbook of Formulas for Emergencies). Journal of Traditional Chinese Medical Sciences 2016;3:1-2. [Crossref]
- Tu Y. Artemisinin-A Gift from Traditional Chinese Medicine to the World (Nobel Lecture). Angew Chem Int Ed Engl 2016;55:10210-26. [Crossref] [PubMed]
- Tu Y. The discovery of artemisinin (qinghaosu) and gifts from Chinese medicine. Nat Med 2011;17:1217-20. [Crossref] [PubMed]
- Yu W, Ma M, Chen X, et al. Traditional Chinese Medicine and Constitutional Medicine in China, Japan and Korea: A Comparative Study. Am J Chin Med 2017;45:1-12. [Crossref] [PubMed]
- Zha LH, He LS, Lian FM, et al. Clinical Strategy for Optimal Traditional Chinese Medicine (TCM) Herbal Dose Selection in Disease Therapeutics: Expert Consensus on Classic TCM Herbal Formula Dose Conversion. Am J Chin Med 2015;43:1515-24. [Crossref] [PubMed]
- Chen Z. Fu Ren Da Quan Liang Fang. China: Song.
- Wang K. Zheng Zhi Zhun Sheng: Ba Ce. China: Ming; 1602.
- Jia X, Zheng Z, Huang Y, et al. Ideas and methods of fresh herb basic research based on constituent structure theory. Zhongguo Zhong Yao Za Zhi 2011;36:2595-8. [PubMed]
- Yang C, Merlin D. Can naturally occurring nanoparticle-based targeted drug delivery effectively treat inflammatory bowel disease? Expert Opin Drug Deliv 2020;17:1-4. [Crossref] [PubMed]
- Mu J, Zhuang X, Wang Q, et al. Interspecies communication between plant and mouse gut host cells through edible plant derived exosome-like nanoparticles. Mol Nutr Food Res 2014;58:1561-73. [Crossref] [PubMed]
- Ju S, Mu J, Dokland T, et al. Grape exosome-like nanoparticles induce intestinal stem cells and protect mice from DSS-induced colitis. Mol Ther 2013;21:1345-57. [Crossref] [PubMed]
- Zhang M, Viennois E, Prasad M, et al. Edible ginger-derived nanoparticles: A novel therapeutic approach for the prevention and treatment of inflammatory bowel disease and colitis-associated cancer. Biomaterials 2016;101:321-40. [Crossref] [PubMed]
- Yang C, Zhang M, Lama S, et al. Natural-lipid nanoparticle-based therapeutic approach to deliver 6-shogaol and its metabolites M2 and M13 to the colon to treat ulcerative colitis. J Control Release 2020;323:293-310. [Crossref] [PubMed]
- ung J, Yang C, Collins JF, et al. Preparation and Characterization of Ginger Lipid-derived Nanoparticles for Colon-targeted siRNA Delivery. Bio Protoc 2020;10:e3685.
- Wang X, Zhang M, Flores SRL, et al. Oral Gavage of Ginger Nanoparticle-Derived Lipid Vectors Carrying Dmt1 siRNA Blunts Iron Loading in Murine Hereditary Hemochromatosis. Mol Ther 2019;27:493-506. [Crossref] [PubMed]
- Zhang M, Xiao B, Wang H, et al. Edible Ginger-derived Nano-lipids Loaded with Doxorubicin as a Novel Drug-delivery Approach for Colon Cancer Therapy. Mol Ther 2016;24:1783-96. [Crossref] [PubMed]
- Song H, Canup BSB, Ngo VL, et al. Internalization of Garlic-Derived Nanovesicles on Liver Cells is Triggered by Interaction With CD98. ACS Omega 2020;5:23118-28. [Crossref] [PubMed]
- Teng Y, Ren Y, Sayed M, et al. Plant-Derived Exosomal MicroRNAs Shape the Gut Microbiota. Cell Host Microbe 2018;24:637-52.e8. [Crossref] [PubMed]
- Golovina EA, Hoekstra FA, Hemminga MA. Drying increases intracellular partitioning of amphiphilic substances into the lipid phase. Impact On membrane permeability and significance for desiccation tolerance. Plant Physiol 1998;118:975-86. [Crossref] [PubMed]
Cite this article as: Yang C. Plant-derived lipid nanoparticles: a new potential therapeutic approach for traditional Chinese medicine’s fresh herbs. Longhua Chin Med 2021;4:19.

